The Oxygen Apocalypse: how symmetry saves us from the flames

📖 20 min read • Cyanobacteria captured light to break a quantum law, unpicking the electron pair, and extinguished ancient life in the process
An artist’s impression of Earth in the early Archean with a purple hydrosphere. Oleg Kuznetsov [CC BY-SA 4.0]
That everything is on fire, slow fire, and we’re all less than a million breaths away from an oblivion more total than we can even bring ourselves to even try to imagine…
— The Pale King, David Foster Wallace
Diradical by Nature
We are protected at every moment by a quantum mechanical safety catch. The most dangerous element of our atmosphere normally can’t react with us. It is physically verboten. Oxygen doesn’t immediately burn our organic matter to a crisp because our biomolecules are a different spin state to O2.
An insuperable barrier of electronic symmetry is our shield (expand the briefing below). Technical Briefing: The Quantum Safety Catch The anatomy of a forbidden reaction — The two \(\pi^{*}\) anti-bonding orbitals of O2 are shown. As the frontier orbitals, these determine the reactivity of oxygen. Two electrons of the same spin can’t occupy the same orbital, so nature has three choices. The same-spin triplet state option (\(^3\Sigma_g^-\)) that makes up 20% of our atmosphere has its reactivity “locked”; the diradical spin density restrained from reacting with typical matter due forbidden spin-interaction rules between molecules. For the opposite-spin paired singlet state options (\(^1\Delta_g\) & \(^1\Sigma_g^+\)), both are highly reactive, but the rarity of the latter attests to a large energy penalty to accessing higher electron spin states. Essentially, “regular” oxygen has two electrons ‘trapped’ in parallel spins, which can’t ‘break into’ regular molecules to cause a reaction — all their orbitals are filled with paired electrons. ▶ Technical Briefing: The Quantum Safety Catch
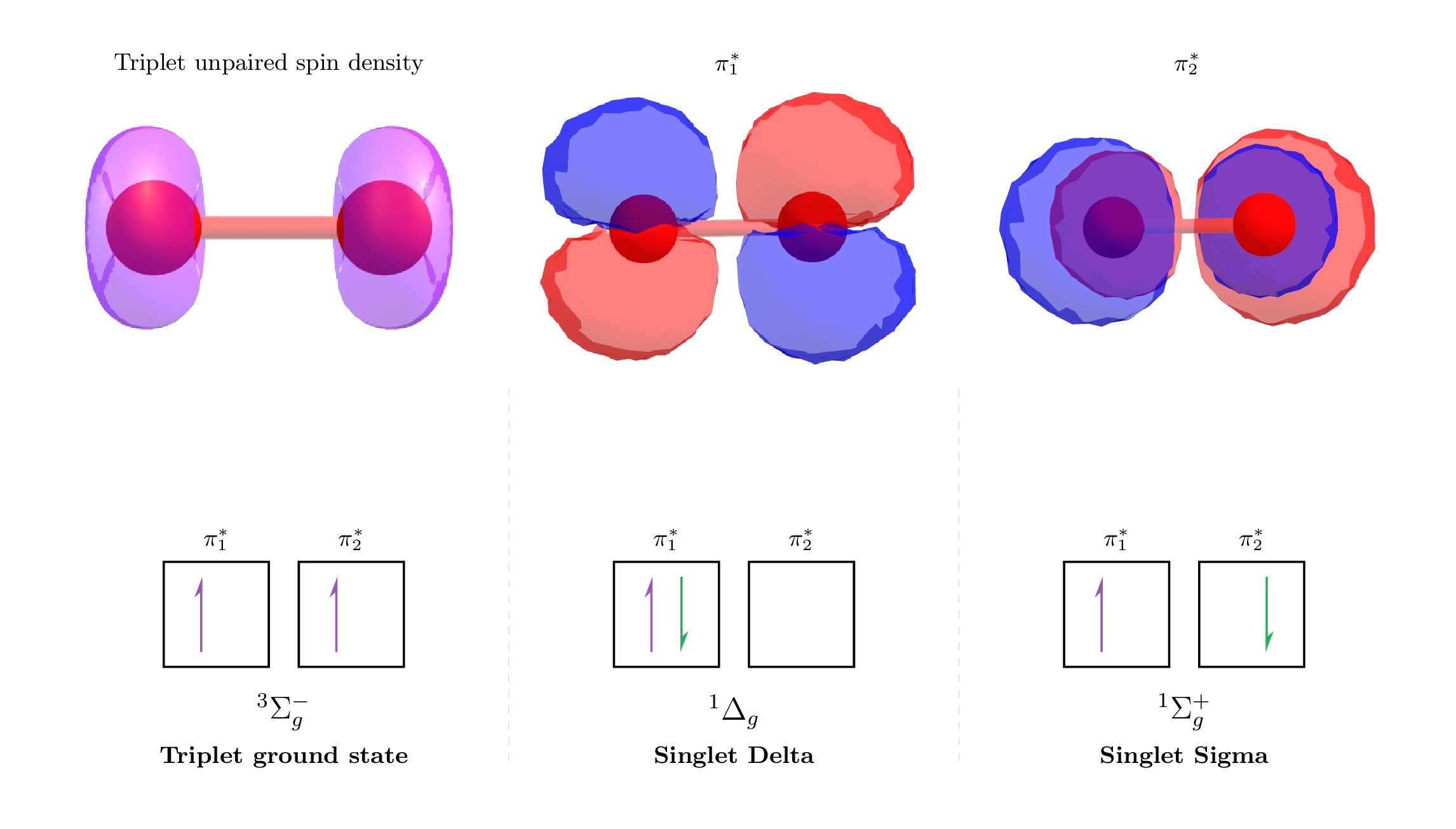
The molecular oxygen in the air is a diradical — it has two unpaired electrons: •O–O•. That makes it a molecule in a ‘triplet’ state. For almost all other molecules on earth, they live in a ‘singlet’ state where all electrons are ‘paired’ off to avoid opening a new shell. They can’t react with each other because doing so would mean the total angular momentum wouldn’t be conserved. This safety catch prevents the atmosphere from incinerating the biosphere.
Singlet oxygen is its triplet twin let loose. Be afraid
They captured the sun…
Light harvesting extremophiles, Chroococcidiopsis thermalis, that can live off lower energy light than most cyanobacteria. T. Darienko [CC BY-SA 4.0]
Before we had the green world as we know it, lifeforms were reliant on chemically ‘reducing’ conditions. Among the first inhabitants of the seas were the archaea (literally, “the ancients”). Some species of ancients could power themselves from abundant hydrogen and carbon dioxide, emitting methane into the atmosphere. Others eked out a diet from sulfur sources. This was the hypothesised Purple Earth, and was fuelled by a chemical environment fundamentally different from our own for billions of years. Until, in the late Archean eon, the first single-cellular shoot of green appeared.
A biological cousin had become a stranger — they hoisted an antenna of chlorophyll towards the sun. The archaea had their own antenna, a simple retinal light gate: it used the centre of the solar spectrum to power primitive pumps. There had already been new life among the ancients; early bacteria, still sulfur dependent, still purple, had begun using a new proto-antenna, but still coexisting with the archaea without threat — the seas were peacefully doubly purple. The chlorophyll antenna of the green bacterial newcomers, in contrast, could harvest the high energy blue and deep red spectral bands. Forced to occupy the solar fringes as the centre was taken, they had hit upon a more potent use of the distance-dimmed energy radiating off the fusion furnace they orbited. They had developed the technology to generate high-voltage electrons from the sun’s rays. The Earth would never be the same again.
…and began to exhale
These chlorophyll wielding bacteria were alien — living in ways previously inaccessible. They could make energy from the sun, they were photo-synthesisers. But their industrious photosynthesis leaked onto a globe a new alien molecule: oxygen gas. O2 had never been a component of the atmosphere before, it was a highly exotic molecule only created in brief flashes when extreme UV radiation smashed apart water or CO2. This new gas was to eventually snuff out the ancients.
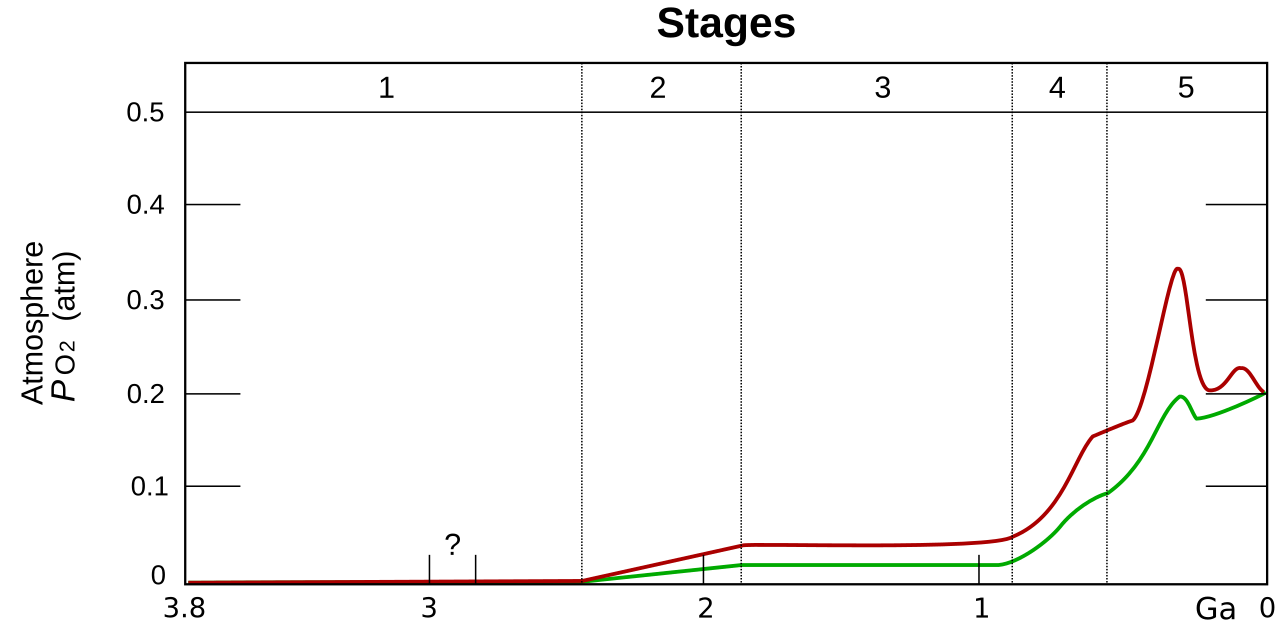
The upper (red) and lower (green) bounds on the oxygen levels in Earth’s atmosphere, on a billions of years timescale. Heinrich D. Holland [CC BY-SA 3.0]
The ancients were hydrogen sulfide metabolisers. H2S could be readily chowed down as an energy source — it fell apart within the cell at ordinary temperature, its hydrogens surrendered as easy fuel. But oxygen was harder to crack: it was locked in a molecular safe, water’s O–H bound 100 kJ/mol more strongly than S–H down a period. H2O required photon-driven electrical impulses and quantum trickery to unpick its bonds.
Enter the cyanos.
Cyanobacteria had staged a metabolic coup with the light energy from their more powerful antenna. The cyanos departed from their ancient ancestors who were constrained to metabolic sulfur sources. For the cyanos, the abundant water they were suspended in became food. They spread anywhere light could reach and began to split the most plentiful fuel source on the planet.
…and the sea boiled
These green cyanobacteria had developed spin-forbidden alien technology. Their oxygen evolving complex (OEC) would radically terraform the Earth. At its heart: a distorted cube of manganese and oxygen, with a jagged edge of calcium. This was their ‘quantum lockpick’ to access the forbidden triplet states. Driven by light power, an ‘S-state’ clock pumped charges from the manganese core. Once electrons were stripped from the core, it became charged enough to fire—rapidly ripping H2O apart into the fuel particles for the cyanobacteria. Technical Briefing: The Quantum Lockpick [Left] The Oxygen Evolving Complex (OEC): “Mimicking the Oxygen-Evolving Center in Photosynthesis”, Chen, Y.; et al., Frontiers of Plant Science, 13, #929532, 2022. Nestled within photosystem II at the start of the photosynthesis machinery is the oxygen evolving complex (OEC) — the ‘lockpick’ that splits apart water to release the protons and electrons used to power the cell. A photon from sunlight knocks an electron off a set of four chlorophyll antennae (P680). To fill the leftover electron hole, an electron is shuffled off the OEC, progressively raising the charge of the manganese (Mn) metals. This light driven process pushes the OEC up the charge states (the “S-clock” cycle). Eventually the OEC is pushed to its limits; from the fourth (S4) state water is ripped apart, pushing the cluster back to resting charge and resetting the S-clock. ▶ Technical Briefing: The Quantum Lockpick
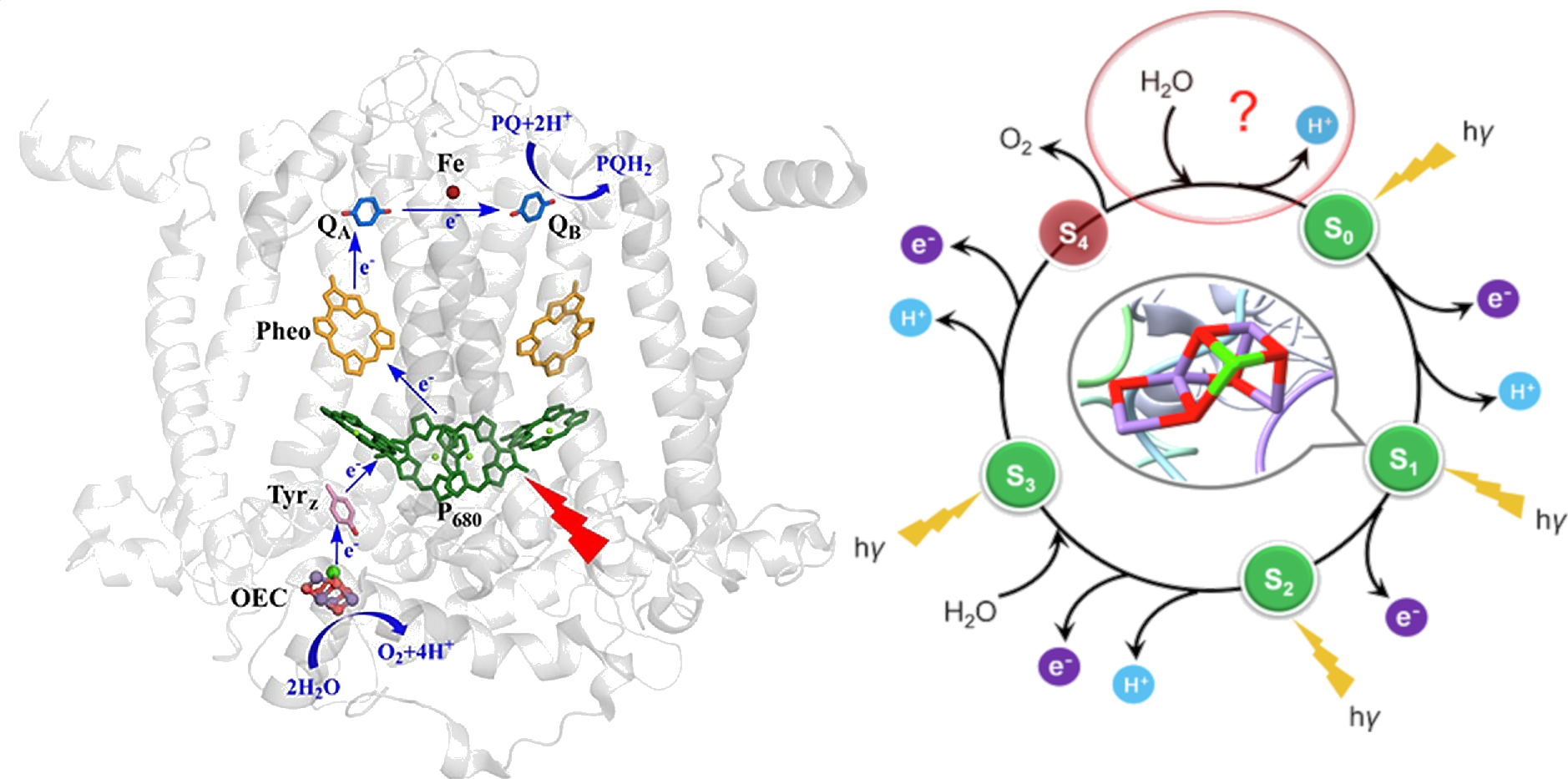
[Right] The “spin diode” S-clock that enables the generation of triplet oxygen from water: “Closing Kok’s cycle of nature’s water oxidation catalysis”, Guo, Y.; et al., Nature Communications, 15, #5982, 2024
▶ Technical Briefing: The S-clock Timebomb
The S-clock is not set to zero. It is self-priming—set one tick ahead to S1. Rather than the clock resting in a ‘ground-state’—where all manganese metals hold their stable electron count—electrons are transferred out of the cluster to tyrosine residues placed nearby. The oxygen evolving complex is capable of self-priming in the complete dark: a powerful cluster of manganese ions that have already been destabilised from their +II configuration by donating electrons to the cluster oxygens and surrounding protein. A primed cannon to bring forth the apocalypse of a furnace world.
Lying in wait until morning, when the next bombardment of photons can push the cluster to higher and higher charge states, and pass a redline. Some propose a ‘firing pin’ manganese ion reaches an outrageous +V charge in the S4 state. Detailed computational work suggests the water molecules are progressively deprotonated and S4 creates an ‘armed bullet’ of an oxyl radical (O\(^{\bullet-}\)). The S4 state is so fleeting not even kilometer-long X-ray Free-Electron Lasers (XFELs) can follow it.
Pushing past S4 triggers a violently reactive reset; ripping four electrons from water to replenish the cluster in femto (10–15) seconds; joining oxo-oxyl groups, each with a missing spin-aligned electron, to form the triplet •O–O• bond, and exhaling oxygen into the environment. The manganese oxide cluster’s magnetic moment acts as a spin template, ensuring the cell doesn’t incinerate itself by creating a singlet. This process is tightly coordinated to avoid creation of cell-destroying peroxide (H2O2) or superoxide (•O2–) species.
The metal cluster core functions as a highly calibrated spin diode. The material for this spin diode was abundant, as free manganese in seawater and mixed with iron in nodules on the ocean floor; a source of “dark oxygen”.

Ferromanganese nodules on the sea floor. North Atlantic Stepping Stones expedition. NOAA, [Public domain]
This new engine has a mineral heart. Nature had left birnessite lying around on the ocean floor — a cubic manganese oxide that catalytically produces oxygen from water in the presence of calcium. As a bubbling rock, birnessite wasn’t a threat to survival. But reconfigured and wielded by an exponentially growing cyano population, the oxygen evolving complex would extinguish ancients and leave behind a furnace world that would come to fuel unrecognisably morphed descendants. Technical Briefing: The Cluster Spin Diode [Left & Middle] The manganese-oxide–calcium cluster of the oxygen evolving complex, and an artificial mimic: “Mimicking the Oxygen-Evolving Center in Photosynthesis”, Chen, Y.; et al., Frontiers of Plant Science, 13, #929532, 2022. The OEC cluster is a distorted manganese-oxide cube, with calcium edge. Though artificial mimics have been created, it is still a somewhat open question if had the OEC had direct mineral origin, and if so, which? Manganese is the natural metal for water splitting due to its versatile oxidation state and favourable redox energetics. Minerals like birnessite also display oxygen evolution, but at far higher overpotentials, and are prone to create reactive oxygen species as side products which would shred a biological cell. The asymmetric cuboid arrangement helps the OEC function as a “spin diode”. The cluster’s four metal centers are in communication to tightly coordinate a series of spin flips which guide the generation of forbidden triplet oxygen. The sequential spin flips are crucial to this near-alien technology developed by the cyanos; they’re what allow the OEC to overcome the—previously unbroken—verboten spin-barrier. ▶ Technical Briefing: The Cluster Spin Diode
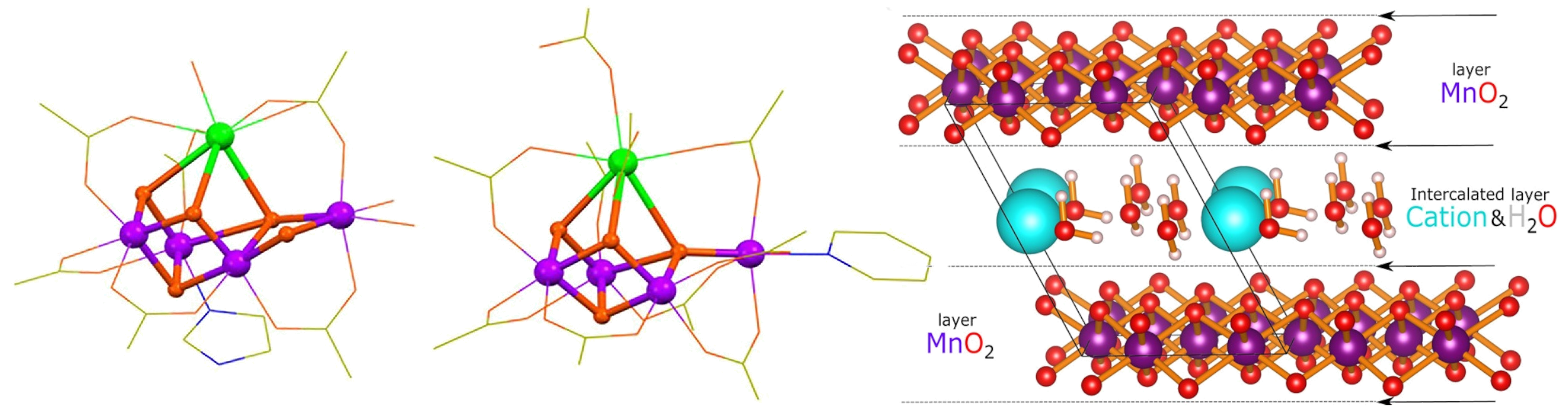
[Right] The natural mineral birnessite that generates oxygen from water in the presence of calcium: “Birnessite: A Layered Manganese Oxide To Capture Sunlight for Water-Splitting Catalysis”, Lucht, K.; Mendoza-Cortez, J., The Journal of Physical Chemistry C, 119, 22838–22846, 2015
The cyanobacteria had broken nature’s rule of closed-shell ground states, pushing through forbidden symmetry regimes with electromagnetic clockwork. From captured spiralling lightwaves the flow of energy was controlled and constrained; the spin gate of the asymmetrically distorted cluster ensured departing electrons would pass through the spin-state barrier. The cyanos could take the substance the world was drowning in —singlet water— and, at ambient temperature, pump out triplet oxygen gas. The cyanos held a quantum lockpick and could now consume the previously forbidden.
…and the oceans bled
There had been other microbial light-eaters, but their atmospheric and oceanic impact was marginal, since previous phototrophs still took electrons from limited dissolved material — hydrogen sulfide or Fe2+ (consumed by the photoferrotrophs). The cyanos, in contrast, could on their own unbalance terrestrial feedback loops, rapidly proliferating away from specific chemical niche locales. While their S-clocks ratcheted through firing cycles at a velocity never before accessed in sun-driven technology, barely any oxygen initially escaped to the atmosphere. The build up in the air was delayed for nearly two billion years. The oceans had a natural buffer to the exotic spin toxin — they contained iron.
Instead of terraforming the atmosphere, oxygen reshaped the oceans first. The oceans held vast reserves of dissolved Fe2+ (making them a vibrant green), that formed iron-hydroxide minerals when oxidised to rust-brown Fe3+. Steadily, the buffer iron was worn through, the bacteria (both oxygen-forming and ferrotrophic) shifted geological masses of dissolved oceanic iron into solids in the form of banded iron formations in the Precambrian.

Banded iron formation in Soudan Underground Mine State Park, MN. James St. John, [CC BY 2.0]
By unpicking oxygen from its water surrounds, bacteria had begun to inexorably rust the oceans. The iron deposited in solid scabs, layered in puff-pastry sheets of blood red and dull metal hues. Rolled, pressed, and folded by the tectonic slippage below.
The Hamersley Range in the Pilbara region of Western Australia preserves some of the most arresting evidence of this process; resurfaced testaments to this planetary-wide haemorrhaging.

Banded iron formation in the Hamersley Range, WA. Dated from the Neoarchaean to the Paleoproterozoic era. James St. John, [CC BY 2.0]
During this purge of iron from the oceans the sea surface was encrusted with ore layers up to a kilometer thick. We use the scabs of the oxidised ferrous sea to build out steel reinforced cities today.
…and their kin choked
The oceans, now cleansed to a clear blue-green, could no longer hold back the corrosive gas. It escaped into the atmosphere and across the globe, wearing down anything that had not evolved chemical defenses.

Diffusing across the skies and into ecological niches, •O–O• oxidised anything it contacted. Seeping through cell walls, oxygen triggered radical chain reactions that destroyed the membrane’s fatty layer, tore through DNA, and ripped apart protein machinery. Microbes unprepared for oxygen were suffused with this alien gas and erased from the genetic pool from the inside. Life had to develop new machinery to cope, or die. Survivors adapted patchwork protection against this molecular cataclysm, imperfect oxygen disposal mechanisms that provided enough antioxidant shielding to outrun extinction. These emergency responses were written into their descendants’ DNA.
As atmospheric oxygen concentration steadily increased, more durable adaptations were required to transcend the chemical danger of this new reality. Some cells found seemingly providential protection under the aegis of oxygen-burning α-proteobacteria. This protection racket would go on to be one of the most successful chemical energetic exchanges in biology: they became mitochondrial powerplants of the incipient multicellular organism, forging previously inaccessible molecular stores of energy. They would come to have domain over this corroding new order.
Most ancients did not survive, only small pockets of anaerobe resilience were left in nooks that were unreachable by this gas. The purple Earth was bleached green by a corrosive spill. With most of the competition extinct, and chemical cycles sundered, the photosynthesisers began to remake the climate.
…and the sky faded
The cyanos had emerged first into a stifling atmosphere, a greenhouse gas layer which was more potent than ours due to high methane (CH4) levels. It was also chemically stultifying before the injection of oxygen, methane lifetimes were in the thousands of years as there was no oxidising radicals to scrub it. The prior bacteria were methanogenic, maintaining this warming blanket. Photosynthesis fundamentally changed this equation.
Within the same carbon budget, CH4 is 30–80x more potent a greenhouse gas than CO2. The millennia-long lifespan of methane was shortened by photosynthetic oxygen to just a decade, converted into carbon dioxide. The linear symmetry of O=C=O leaves it with fewer active infrared absorption modes than CH4: particular geometric distortions that can absorb longwave heat radiating back from the Earth’s surface. The 1:1 exchange of CH4 to CO2 by catalytic oxygen-linked radical reactions had made the greenhouse blanket more translucent to heat. While the oceans had buffered the cyanobacteria from the consequences of their waste gas for billions of years, their emissions destroyed the greenhouse layer in a thousandth of that . There was no human heavy industry emitting gigatonnes of CO2 into the atmosphere.
The sun was dimmer back then. With nothing to compensate, a long winter was coming.
…and the Earth froze

An artist’s impression of “Snowball Earth” during the Huronian Glaciation. Oleg Kuznetsov [CC BY-SA 4.0]
Tipping the Earth’s temperature balance, the cyano’s solvent-fuel-transport wonder substance began to freeze. Glaciers crept from the poles towards the equator, dramatically envisioned as a “snowball Earth” where ice had a stranglehold on the globe. The full extent of the ice after the initial oxidation event is uncertain, likely not a complete encasement, but oxygen had set in motion an unstoppable feedback loop: as the surface reflectivity increased temperatures plunged. The lockpick that allowed cyanos to break down inexhaustible water now caused them to be encased in crystalline cages of it. It was an approach towards self-extinction. But a totality wasn’t reached, the climate oscillated in and out of frozen conditions, life barely clinging on in ice-free oases.
The accumulation of greenhouse gases from the volcanic ruptures during tectonic shifts broke the globe free from this frozen end state. But this thawed world was a new one, the atmosphere had been injected with triplet oxygen, a potent fuel source for any metabolism aggressive enough to handle this incendiary substance.
…and life rose up
The oxygen apocalypse had largely swept aside the Purple Earth of the ancients, though archaea are still detectable in almost every habitat. The exact emergence timelines of various aerobic forms of life in oxic/anoxic and sulfur-dominated conditions are not completely clear-cut. But lifeforms would never be the same, eukaryotes —complex, and eventually multicellular, life— could emerge since there was more energy to work with. The trees in our parks were to be descended from the dominant cyanos, the oxygen evolving complex to be incorporated into the chloroplast of every leaf. While timepoints recording last and first common ancestors are still estimated in millions-of-years ranges, the outcome was inevitable: life would be oxygen-breathing, rocketed into fierce metabolic competition.
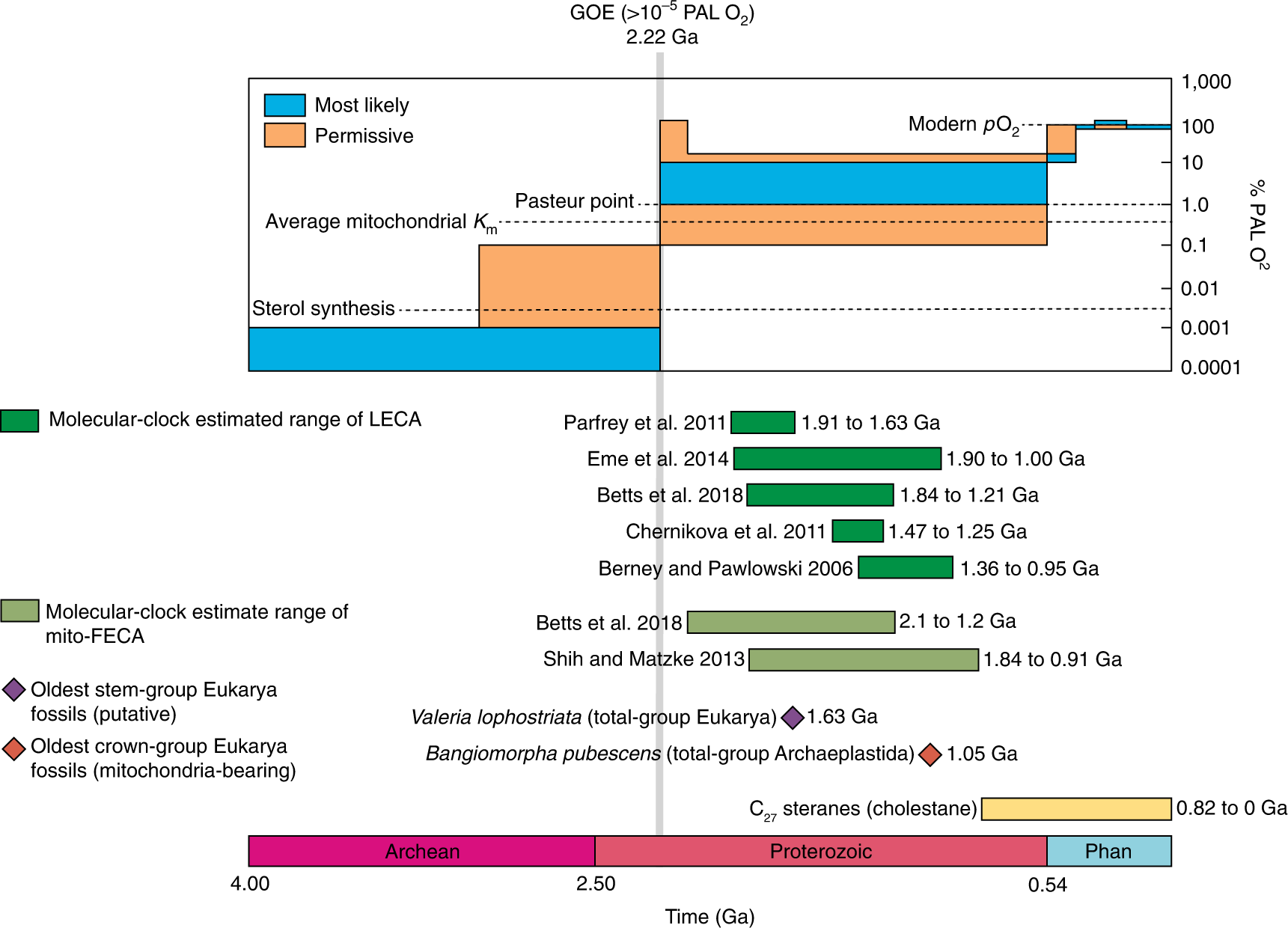
Molecular, geological and fossil evidence for the arrival of complex life following the Great Oxidation Event (GOE). “Eukaryogenesis and oxygen in Earth history”, Mills, B.; Boyle, R.; Daines, S.; Sperling, E.; Pisani, D.; Donoghue, P.; Lenton, T.; Nature Ecology & Evolution, 6, 520–532, 2022
…and aged away
Far from being a rejuvenating substance or a breath of fresh air, the cellular combustion of triplet oxygen is a corrosive bargain against the clock. Addition of a single electron to •O–O• during a biochemical process gone awry creates the superoxide radical (•O2–) which is not as strictly spin-forbidden against reacting with regular matter. This is the opening slippage in oxygen control that unshackles a cascade of other reactive oxygen species, which tear into biochemical structures and degrade cellular function. Oxidative stresses are one of the major contributors to cellular ageing in all aerobic organisms; the trade for a powerful metabolism is the shortening of cellular lifespan.
The production of these reactive oxygen species is ironically also mediated through iron — the same buffering element precipitated out of the Archean-era ocean by the oxygen evolving complex, now facilitating the accumulation of cellular damage from the OEC’s waste.
The net Haber–Willstätter(Weiss) reaction indicates how absorbed superoxide (•O2–) can create reactive oxygen species. It was co-proposed by Fritz Haber of Haber-Bosch fame in his final paper, published just after his death. The proposal was a breakthrough historical starting point (and a misnomer) but didn’t fully bear out in mechanistic studies. Fenton implicated iron species as the main pathway radical propagation. Direct reaction of superoxide with hydrogen peroxide would actually unleash singlet oxygen.
Entire complex biological networks of antioxidants have been evolved to patch the damage of an oxygen metabolism: from superoxide converting enzymes, to radical scavenging vitamins, and phenolic aromatic compounds that can stabilise radical species into a less destructive form. Life’s chemical potential was turbocharged, but with it comes an accumulation of organic corrosions that has to be carefully managed by patchwork self-immolations until the cell fails.
…and everything’s on fire.
The atmosphere is now a great, slow, combustion engine. The steel girding our housing is smelt from the oxidised blood of the ocean. The plants are descendants of microbes which unpicked the water bond through a photocharged spin diode. They support our every breath, but their ancestors chemically obliterated the Earth. The ancients are all but wiped out wherever oxygen could reach. Our lungs fill with a combustible gas, which only our mitochondria —internalised from an entirely different lineage— can burn with any modicum of control. A spin barrier ensures that this triplet oxygen, contributing the reactive fifth of our atmosphere that allows life to tick on, is not cause for immediate incineration. But now, living under an oxygen-rich sky, under the dictates of a rust-based order, still the law: all things decay and age.
Oooooxxxyyygeeeennnn
— To Be Kind, Swans